温馨提示:本文仅基于疾病科普分享,不能代替医院诊疗。意见仅供参考,具体治疗方式请遵医嘱。
责任编辑:觅健科普君
参考来源:
[1]Qin SK et al., First-line Lenvatinib Plus Pembrolizumab for Advanced Heptocellular Carcinoma: LEAP-002 Asian Subgroup Analysis. JSMO, 2023
[2]Chih-Cheng Tan, Chi-Jung Wu, Pei-Chang Lee, et al, Add-on Gefitinib for Unresectable Hepatocellular Carcinoma after Lenvatinib Failure. APASL 2023 ppp-304.
[3]FDA Grants Orphan Drug Designation to Ymmunobio's CEACAM Antibody for the Treatment of Liver Cancer. News release.
Ymmunobio. February 28, 2023. Accessed March 1, 2023. https://www.ymmunobio.com/post/fda-grants-orphan-drug-designation-to-ymmunobio-s-ceacam-antibody-for-the-treatment-of-liver-cancer.
[4]Janicot M, Rupalla K, Schmidt A, et al. Novel, immune agonistic CEACAM1/5 antibody (YB-200) demonstrates anti-tumor efficacy and significantly increases B-cell response in syngeneic liver Hepa1-6 tumor microenvironment. J Immunother Cancer. 2022;10(suppl 2):A1438. doi:10.1136/jitc-2022-SITC2022.1384.
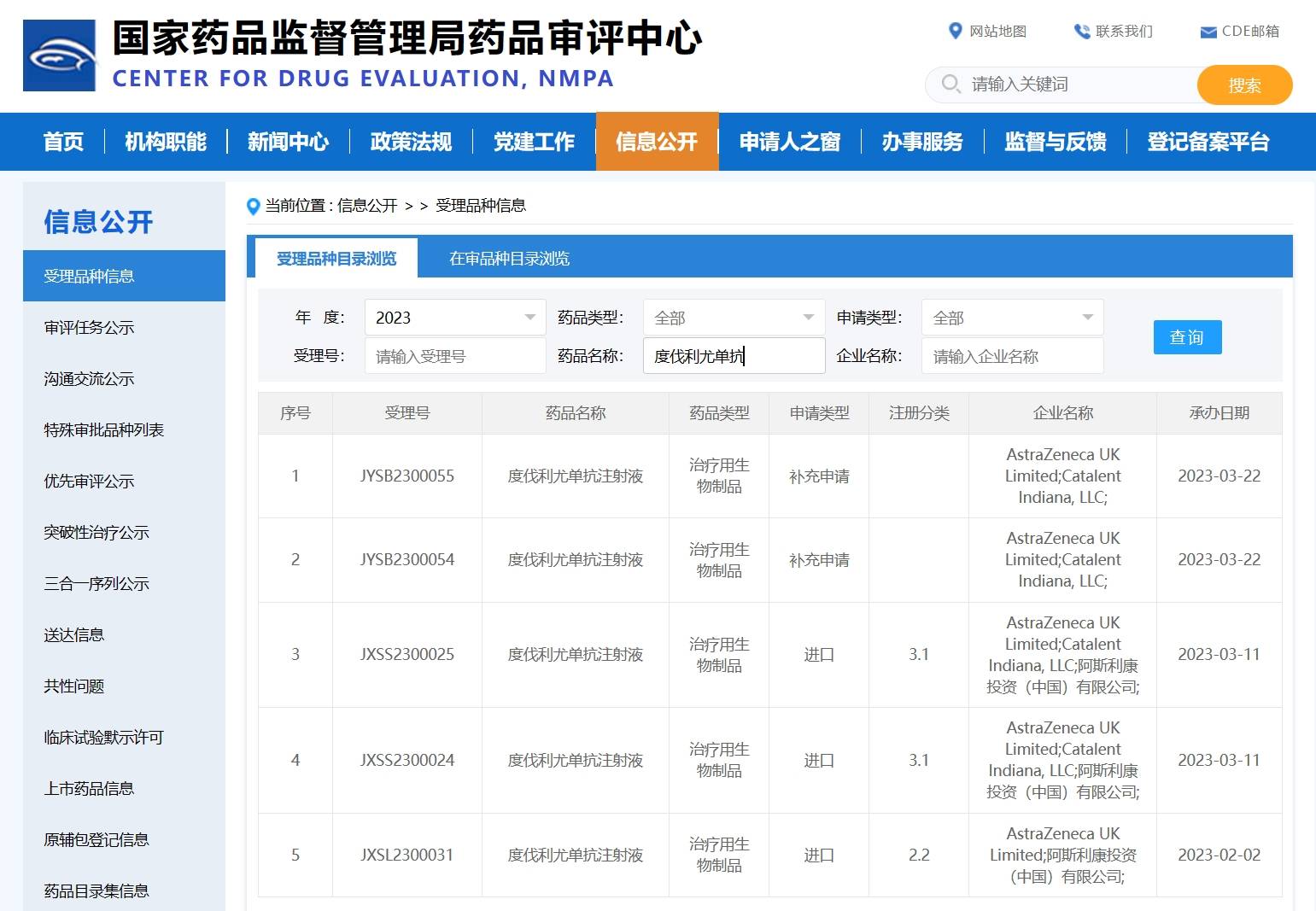
[5] Do-Youn Oh, et al. 78P - Updated overall survival (OS) from the phase III TOPAZ-1 study of durvalumab (D) or placebo (PBO) plus gemcitabine and cisplatin (+ GC) in patients (pts) with advanced biliary tract cancer (BTC). ESMO Asia Congress 2022. Annals of Oncology (2022) 33 (suppl_9): S1454-S1484. 10.1016/annonc/annonc1123.